Benzal Chloride: Properties, Production and Uses
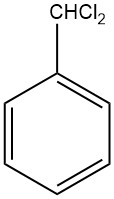
What is Benzal chloride?
Benzal chloride, also known as dichloromethylbenzene, α,α-dichlorotoluene, or benzylidene chloride, is an organic compound with the chemical formula C6H5CHCl2. It is a colorless liquid that is synthesized by chlorinating the side chain of toluene.
The first synthesis of benzal chloride was reported in 1848 by A. Cahours, who reacted benzaldehyde with phosphorus pentachloride (PCl5).
Table of Contents
1. Physical Properties of Benzal Chloride
Benzal chloride is a colorless liquid with a pungent odor. When exposed to moist air, it emits fumes and can cause strong irritation to mucous membranes and eyes. Its physical properties are as follows:
| Property | Value |
|---|---|
| Molecular weight | 161.03 g/mol |
| Melting point | -16.2°C |
| Boiling point | 205.2°C |
| Density at 20°C | 1.2536 g/cm³ |
| Vapor pressure at 20°C | 0.6 kPa |
| Refractive index (n20D) | 1.5503 |
| Surface tension (σ) at 20°C | 40.1 mN/m |
| Dynamic viscosity (η) at 20°C | 2.104 mPa s |
| Specific heat at 25°C | 222 J mol⁻¹ K⁻¹ |
| Heat of vaporization at 72°C | 50.4 kJ/mol |
| Property | 0°C | 20°C | 30°C | 57°C | 79°C | 135°C |
|---|---|---|---|---|---|---|
| Density | 1.2691 | 1.2536 | 1.2417 | 1.2122 | 1.1877 | 1.1257 |
| Vapor pressure | 0.0 | 0.6 | 0.8 | 1.3 | 1.9 | 4.0 |
| Refractive index | 1.5499 | 1.5503 | 1.5506 | 1.5513 | 1.5523 | 1.5542 |
| Surface tension (σ) | 40.4 | 40.1 | 39.8 | 38.9 | 38.2 | 36.4 |
| Dynamic viscosity (η) | 2.12 | 2.104 | 2.089 | 2.058 | 2.023 | 1.948 |
Benzal chloride can form several azeotropic mixtures, with examples listed in Table 2.
| Component | boiling point, °C | Benzal chloride,wt % |
|---|---|---|
| Isoamyl lactate | 201 | 45 |
| n-Octanol | 195 | ca. 10 |
| Camphor | 210 | 25 |
| Acetamide | 191 | 85 |
| Caproic acid | 199 | 64 |
Benzal chloride is soluble in ethanol, diethyl ether, chloroform, and carbon tetrachloride, but its solubility in water is limited (0.05 g/L at 5 °C and 0.25 g/L at 39 °C).
In ethanolic and aqueous solutions, alcoholysis or hydrolysis reactions may occur.
The solubility of chlorine in 100 g of benzal chloride varies with temperature, being 6.2 g at 30 °C, 4.3 g at 50 °C, and 1.5 g at 100 °C.
2. Chemical Reactions of Benzal Chloride
Benzal chloride can be converted into benzotrichloride by chlorinating agents.
In the presence of Lewis acids, chlorination of the aromatic ring produces chlorobenzal chlorides, which are isomeric compounds.
Benzaldehyde is produced when benzal chloride is hydrolyzed under acidic or alkaline conditions.
Benzal chloride can be polymerized in the presence of compounds such as AlCl3 and FeCl3.
Sodium metal can convert benzal chloride into stilbene.
3. Production of Benzal Chloride
Benzal chloride is produced by chlorinating the side chain of toluene. The same process and plant configuration are used to produce benzyl chloride and benzotrichloride.
However, the molar chlorine-to-toluene ratio must be adjusted to achieve optimal yields. The high-boiling byproduct fraction obtained during benzyl chloride production mainly consists of benzal chloride and benzyl chloride.
This byproduct fraction can be recycled in the synthesis of benzal chloride. As a result, the production of benzal chloride and benzyl chloride are closely intertwined and often operate simultaneously at the same production facility.
4. Uses of Benzal Chloride
Benzal chloride is mainly used to produce benzaldehyde and cinnamic acid. It can be hydrolyzed in the presence of water at temperatures above 100 °C, using either alkaline or acidic agents. Friedel–Crafts catalysts are effective catalysts for this reaction.
They can also be used for mixtures containing both benzyl chloride and benzal chloride. In these cases, it is said that benzyl chloride remains unchanged and only benzaldehyde is produced.
However, this process may not be commercially viable due to the nearly identical boiling points of benzyl chloride and benzaldehyde. The separation of these two compounds through fractional distillation would be prohibitively expensive.
5. Toxicology of Benzal Chloride
Benzal chloride is a colorless liquid with a pungent odor. It emits fumes when exposed to moist air and can cause irritation to the eyes, skin, and mucous membranes.
The available data on the toxicity of benzal chloride indicates that it is less toxic than benzyl chloride. The acute oral toxicity (LD50) of benzal chloride in rats and mice is 3250 mg/kg and 2460 mg/kg, respectively.
The inhalation toxicity of benzal chloride is also lower than that of benzyl chloride, with a LC50 (2-hour exposure) of 400 mg/m³ in rats and 210 mg/m³ in mice.
Benzal chloride is known to be mutagenic and has been associated with the development of skin tumors and lung adenomas in mice. However, there are no separate studies on the carcinogenic effects of benzal chloride in humans.
Therefore, it is classified as a Group 2B carcinogen (evidence of sufficiency in animals, insufficient data in humans) by the International Agency for Research on Cancer (IARC) and as a category 2 carcinogen by the German Research Foundation (DFG).
Reference
- Benzyl Chloride and Other Side-Chain-Chlorinated Aromatic Hydrocarbons; Ullmann’s Encyclopedia of Industrial Chemistry. – https://onlinelibrary.wiley.com/doi/abs/10.1002/14356007.o04_o01.pub2
