Gelatin: Properties, Production and Uses

Gelatin is a translucent, colorless, brittle (when dry), flavorless, edible, multifunctional protein polymer, hot-water extracted from “collagens” obtained from various animal and fish byproducts.
The raw materials used in gelatin production are sourced from various animal sources such as pig skins, bovine hides, cattle and pig bones (ossein), and fish skins.
The parent proteins that give rise to gelatin, known as “collagens,” belong to the collagen family of proteins that make up approximately 30% of the animal body’s total protein content.
Collagens are essential structural proteins found in various connective tissues, including skins, bones, tendons, ligaments, corneas, cartilages, blood vessels, and guts. During the gelatin production process, these collagen proteins are partially hydrolyzed.
The primary materials utilized in the production of gelatin are animal and fish byproducts, including pig skins, bovine hides, cattle and pig bones (ossein), and fish skins.
Table of Contents
1. Production of gelatin
Gelatin is a commercially available protein derived from animal collagen found in skins and bones. The primary sources of raw materials used for gelatin production include cattle hides, bovine and pig bones, and pigskins.
In order to make the collagen soluble in hot water, each raw material requires a specific pretreatment process.
During the pretreatment procedure, the collagen-containing material undergoes a series of changes, such as swelling and softening, cross-linking, and hydrolysis of non-collagenous proteins’ peptide bonds. Additionally, various impurities are extracted from the material.
There are two main types of gelatin: type A, which is produced by acid-pretreated raw materials, and type B, which is produced by alkaline processing.
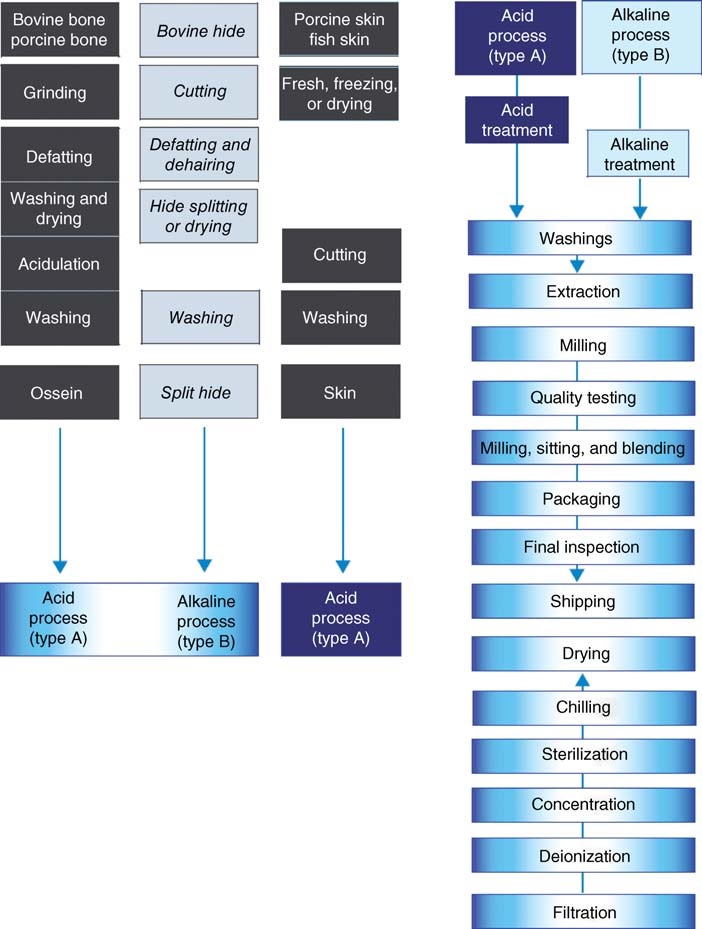
1.1. Acid Process
The acid process is typically used for pigskin and, on occasion, for specific types of ossein. Washed pigskins are treated with diluted mineral acids at low temperatures for approximately 24 hours.
This pretreatment is sufficient to cleave acid-labile peptide bonds present in pigskin collagen. After partial neutralization, the gelatin is extracted with hot water.
The extraction of gelatin usually occurs stepwise, with successive increases in temperature and time. Gelatins from sequential extractions exhibit distinct physical and chemical properties.
The initial extract has the highest gel strength and molecular mass and is very low in color.
Subsequent extractions, obtained at elevated temperatures, contain a higher proportion of low molecular weight peptides, resulting in lower gel strength, and are more intense in color and haziness.
1.2. Alkali Process
Type B gelatin is produced by pretreating cattle hides, bovine, and porcine ossein with alkali, such as lime or sodium hydroxide, at ambient temperature.
The liming process varies from several weeks to several months and removes impurities, cross-links, and peptides, primarily those of non-collagenous origin.
Liming time is a critical factor in further processing. Dilute solutions containing 3-10% gelatin are obtained by hot-water extraction.
The solutions are filtered to remove suspended matter, deionized to eliminate inorganic ions and then concentrated up to 25-35 wt% by ultrafiltration and vacuum evaporation, filtred and sterilized.
The concentrated solution is swiftly cooled to form a gel and extruded as noodles, which are placed on a stainless steel net to dry with hot, clean air.
The net passes slowly through a drying chamber with numerous zones that have controlled temperature and humidity. The typical temperature range varies from approximately 30°C to 70°C in the final zone.
The dried gelatin, with a moisture content of around 10%, is broken and milled.
2. Chemical properties of Gelatin
During gelatin production, the animal raw material is subjected to either dilute acid (type A process) or alkali (type B process) treatment to partially cleave the interchain cross-links that define the thermal tolerance of collagen fibrils.
This breakdown results in the formation of “hot water-soluble collagen,” or gelatin, which is extracted with hot water that melts out the collagen fibrils into their individual α-chains.
Gelatin is a polydisperse mixture of protein fragments of varying molecular masses, ranging from 15,000 to >400,000 Dalton.
Its composition depends on the level of chemical/thermal hydrolysis of the α-chains and the level of interchain cross-link hydrolysis. Except for glutamines and asparagines, its amino acid composition corresponds to that of the post-translationally processed collagen from which it is derived.
Acid- and base-catalyzed deamidation reactions convert some glutamines/asparagines for a type A-processed gelatin and most of the glutamines and almost all asparagines for a type B-processed gelatin into their acidic counterparts.
Gelatin derived from porcine or bovine raw material typically contains a minimum of 11% hydroxyproline, which is used for identification and quantification purposes.
Quantitative analysis of gelatin shows about 50.5% carbon, 25.2% oxygen, 17.0% nitrogen, and 6.8% hydrogen. The ash content varies depending on the origin of the raw material and the method of processing.
Pigskin-derived gelatin contains small amounts of chlorides and sulfates from acid treatment before extraction, while bone and hide-derived gelatin contain calcium and sodium salts from the lime used in pretreatment.
Dry gelatin stored in airtight containers at room temperature remains stable for long periods, but degradation may occur in solution due to extremes of pH, temperature, and proteolytic enzymes like papain or trypsin.
Gelatin can be chemically modified to alter its properties, such as through acylation, carbamylation, succinylation, phthalation, or carbamylation. These modified products are used for specialized pharmaceutical and photographic applications.
Methacrylated gelatin is a new derivative that is suitable for technical applications. Gelatin can also be permanently cross-linked by reacting it with aldehydes such as formaldehyde, glyoxal, or glutaraldehyde.
3. Uses of Gelatin
Gelatin, although primarily a foodstuff, has been valued not only for its nutritional contribution but also for its functional properties. Its applications extend beyond the food industry, and are important in the pharmaceutical, cosmetic, photographic, and technical fields.
Due to its molecular structure and structure-function relationship, gelatin exhibits a range of properties, including gel formation, water binding, thickening, emulsion formation and stabilization, foam formation and stabilization, film formation, adhesion/cohesion, protective colloid function, and inhibition of ice-crystal formation.
3.1. Food Industry
In the food industry, edible gelatin and gelatin hydrolysate are commonly used ingredients for the production of various food products.
Different properties and applications require the production of specific gelatin types.
Leaf gelatin is widely used for household purposes due to its ease of measurement.
Special instant gelatins are available for preparing cream cakes and desserts without heating, while powdered gelatin and gelatin hydrolysate are used in the processing of confectionery, dairy products, desserts, meat products, beverages, and more.
3.2. Pharmaceutical Applications
In the pharmaceutical industry, gelatin is widely used for the manufacture of soft and hard capsules, as a bulking agent, for coating water-insoluble vitamins, as a primary component for surgical sponges, and as a binder in tablets.
Gelatin is also recommended as a food supplement for reducing joint pain, improving bone health, and enhancing skin beauty.
3.3. Cosmetics
Gelatin and its hydrolysates are commonly used in skin and hair-care products due to their nontoxic and non-irritant nature.
In skin-cleansing preparations, they reduce the potential for irritation caused by anionic surfactants.
In hair-care preparations, gelatin hydrolysates protect the structure of hair and minimize damage to hair structure and biochemistry caused by permanent waving, bleaching, or coloring.
3.4. Photography
Gelatin is an essential component in photographic materials, filling up a major part of photo-emulsion films.
Its unique characteristics, such as sol-gel transformation, interactivity with silver ions, cross-linkability, and water swell-ability, make it an indispensable ingredient for the development of latent images and dye formation during photographic material processing.
Reference
- Gelatin; Ullmann’s Encyclopedia of Industrial Chemistry. – https://onlinelibrary.wiley.com/doi/10.1002/14356007.a12_307.pub2
